Detailed Description
Collection Statistics |
|
|---|
Modalities | MR, SEG | Number of Participants | 222 | Number of Studies | 847 | Number of Series | 9032 | Number of Images | 386,528 | | Images Size (GB) | 76.2 |
Requirements for MR imaging (As specified in the ACRIN 6657 protocol )Imaging time points: MRI exams were performed within four weeks prior to starting anthracycline-cyclophosphamide chemotherapy (T1, MRI1), at least 2 weeks after the first cycle of AC and prior to the second cycle of AC (T2, MRI2), between anthracycline-cyclophosphamide treatment and taxane therapy if taxane was administered (T3, MRI3), and after the final chemotherapy treatment and prior to surgery (T4, MRI4). The study schema is shown in Figure 1 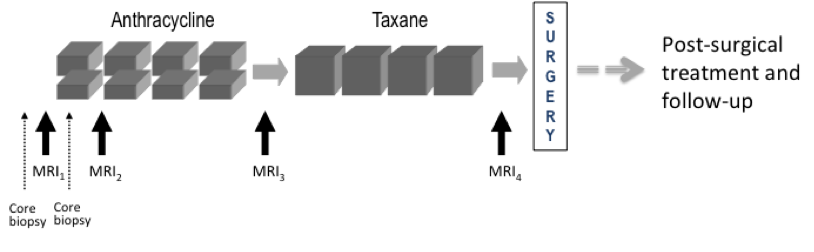
Figure 1. CALGB 150007 and ACRIN 6657 study schema. Imaging protocol: MR imaging was performed on a 1.5 Tesla field strength scanner using a dedicated breast radiofrequency coil. The image acquisition protocol included a localization scan and T2-weighted sequence followed by a contrast-enhanced T1-weighted series. All imaging was performed unilaterally over the symptomatic breast and in the sagittal orientation. The contrast-enhanced series consisted of a high resolution (≤1mm in-plane spatial resolution) three-dimensional, fat-suppressed, T1-weighted gradient echo sequence with TR≤20 ms, TE = 4.5 ms, flip angle ≤ 45º, 16-18 cm field-of-view, minimum matrix 256x192, 64 slices, slice thickness ≤ 2.5 mm. Scan time length for the T1-weighted sequence was required to be between 4.5 and 5 minutes. The sequence was acquired once before contrast injection and repeated at least twice following injection. Tumor diameter measurement and volumetric analysis: Tumor longest diameter (LD) was measured by the site radiologist as the greatest extent of disease on baseline MR images, including intervening areas of non-enhancing tissue. The same measurement direction was used on all subsequent MRI exams. The primary predictor variable, functional tumor volume (FTV) was measured from contrast-enhanced images using the signal enhancement ratio (SER) method. Volumetric analysis, including Quality Control assessment, was performed centrally at the Breast Imaging Research Program (BIRP) laboratory at University of California at San Francisco (UCSF). Detailed information about the DICOM data is available in the DICOM Dictionary . Further information on these studies can be found at:Imaging Data Transfer HistoryThe processing of the MR image data for ACRIN 6657 consisted of the following steps between image acquisition and the creation of this shared data set on TCIA: - MRI studies were sent from the study centers to the ACRIN Core Lab either via media (DVD) or the TRIAD program
- Image data were de-identified and centrally archived at the ACRIN Core Lab
- Archived data was sent to the Breast Imaging Research Program (BIRP) at the University of California, San Francisco (UCSF) for volumetric analysis.
- De-identified image data, derived analysis maps and segmentations, and ancillary data files were transferred from UCSF to TCIA for data sharing.
While every effort was made to preserve the integrity of both the original image data and image meta-data (DICOM attributes, public and private), multiple file transfers and strict adherence to HIPPA guidelines for patient confidentiality may have resulted in loss of some data. If any questions arise, or patient PHI is found in any data on this collection, please contact the UCSF Breast Imaging Research Program (BIRP). For scientific or other inquiries about this dataset, please contact TCIA's Helpdesk. Curated Data SetsIn addition to the complete set of ACRIN 6657 imaging studies ("Level 0" data), the following curated data sets based on UCSF QC assessment, protocol compliance and data completeness are provided: - Level 1: MRI Longest Diameter (LD)
- Level 2a: SER Volume Dataset for ongoing volumetric analyses (updated 9/17/16)
- Level 2b: SER Volume Dataset for pCR Analysis (Hylton, et al; Radiology 2012)
- Level 3: SER Volume Dataset for RFS Analysis (Hylton, et al; Radiology, 2016)
The image data sets are accompanied by Excel files with selected patient clinical and outcome data. Data subset Descriptions
Data set | subjects | All Series | DCE + Derived Only | DCE Only | Clinical and outcome data |
|---|
| Level 0: Complete image data set | 222 | | NA | NA | | | Level 1: Studies with MRI LD measurements | 219 | | NA | NA | | | Level 2a: Studies with SER Volume measurements | 207 | | | | | | Level 3: Studies used in primary aim analysis | 162 | | | | |
Level 0: Complete I-SPY 1 / ACRIN 6657 MRI DatasetThis data set is comprised of all HIPPA compliant, DICOM compliant MRI series. Level 0 image data set consists of 847 on-study MRI studies on 222 subjects in the UCSF image database.
One patient in the image data collection (I-SPY ID 1079 ) does not appear in the Feb. 2, 2011 I-SPY FINAL LOCKED clinical data dump. So no clinical or outcome data is available for this subject. Level 1: MRI exams for which longest diameter was measuredThis data set is comprised of all studies with MRI measured longest diameter (LD) values reported.
839 MRI studies have LD reported in the I-SPY 1 clinical database, of which 5 studies are not present in either the UCSF or ACRIN image data collections (see Table 1).
Level 1 image data set consists of 834 MRI studies on 219 subjects in the UCSF image database
Table 1. Studies that have LD measurement but are missing from the UCSF and ACRIN TRIAD image data collections: |
|---|
ID 1071, T1
ID 1138, T1 | ID 1101, T3
ID 1040, T4 | ID 1187, T4 |
Level 2a: Good SER Volume Dataset – updated 9/3/14, 9/17/16This data set is comprised of the patient studies which, following quality reviews in 2014 and 2016, were judged to have sufficiently good image quality and protocol compliance for volumetric DCE SER analysis. Rejection criteria included: incomplete volumetric DCE acquisitions, lack of a 2nd post-contrast acquisition, variability in fat suppression across the image, observed patient motion during the DCE acquisition, significant DCE protocol deviations such as changing scan parameters or image position during DCE acquisition. Level 2a image data set consists of 706 MR studies on 207 subjects in the UCSF image database. These include 7 studies not included in Level 1 (no MRI LD recorded) as listed in Table 2.
Table 2. Studies in Level 2a (good volumetric analysis) that do NOT have LD measures: |
|---|
ID 1059, T4
ID 1079, T2 *
ID 1104, T4 | ID 1192, T2
ID 1212, T4 | ID 1215, T1
ID 1238, T2 | - Patient 1079 does not appear in the Feb. 2, 2011 I-SPY FINAL LOCKED clinical data set. So no clinical or outcome data is available.
|
Level 2b: SER Volume Dataset Reported in Hylton et al. (Radiology, 2012) *This data set is comprised of the patient studies analyzed for pCR outcome and reported in the 2012 Radiology paper on ACRIN 6657 pCR results *. This data set is not provided as a shared list, as it is not recommended for use in further analysis. It is described here because it is the data set from which the Level 3 (primary aim analysis) set was derived. Inclusion and exclusion was determined by quality and protocol reviews available at that time. In addition to the exclusion criteria listed for Level 2a, studies done with imaging in the axial plane, in violation of the sagittal orientation specified in the trial imaging protocol, were excluded due to processing limitations of the analysis software. Similarly, bi-lateral sagittal acquisitions (alternating left and right volumetric acquisitions) were excluded. Level 2b image data set consists of 707 MRI studies on 207 subjects in the UCSF image database. - Hylton, N. M., Blume, J. D., Bernreuter, W. K., Pisano, E. D., Rosen, M. A., Morris, E. A., Weatherall, P. T., Lehman, C. D., Newstead, G. M., Polin, S., Marques, H. S., Esserman, L. J., & Schnall, M. D. (2012). Locally Advanced Breast Cancer: MR Imaging for Prediction of Response to Neoadjuvant Chemotherapy—Results from ACRIN 6657/I-SPY TRIAL. https://doi.org/10.1148/radiol.12110748 PMC3359517
Tables 3 and 4 show the specific inclusion/exclusion differences between Levels 2a and 2b:
Table 3. 16 studies accepted for SER analysis since 2008 (in Level 2a but not in 2b) |
|---|
ID 1005, T3
ID 1043, T2
ID 1046, T4
ID 1057, T3
ID 1074, T3
ID 1084, T1 | ID 1110, T4
ID 1139, T4
ID 1159, T4
ID 1176, T2
ID 1201, T2 | ID 1203, T4
ID 1206, T4
ID 1225, T3
ID 1219, T3
ID 1228, T4 |
| Table 4. 17 studies rejected since 2008 (in Level 2b but not in 2a) | | Study ID and TP | Reason for rejection for volumetric SER analysis (level 2a) |
|---|
1007 T4 * | No fatSat; Different protocol from T1 | | 1035 T4 * | Only 1 post scan then acq. parameters changed | | 1045 T1 | Alternating laterality acquisitions, 2 minute time gap | | 1047 T1 * | Image position changed during DCE | | 1053 T2 * | Alternating laterality acquisitions, bad pre- acquisition | | 1053 T4 * | Alternating laterality acquisitions | | 1055 T1 * | Alternating laterality acquisitions, 4 minute time gap | | 1086 T1 * | Alternating laterality acquisitions, time gap, different protocol from T4 | | 1091 T1 * | Changing acq. parameters during DCE | | 1095 T2 * | Only 1 post scan then acq. parameters changed | | 1173 T3 * | Off protocol timing | | 1206 T1 | Bad DCE timing, 20 minute delay | | 1206 T2 | Bad DCE timing, 1'29" acquisition time | | 1224 T3 * | Scan position changed during DCE | | 1230 T3 * | Scan position changed during DCE | | #128 T1, T2 | 2 studies for ineligible patient: ACRIN ID 128 (no I-SPY ID) | - Subjects that were included in the primary aim analysis (Level 3)
|
Level 3: Subset of Level 2b used in primary aim analysis, reported in Hylton et al. (Radiology, 2016) *This data set is comprised of the patient studies analyzed for RFS outcome and reported in the 2015 Radiology paper on ACRIN 6657 survival results (Hylton et al, Radiology *). Table 5 shows the 45 patients excluded from the level 2a cohort for this analysis. Please see the publication for specific information on exclusions of patients from this group. Level 3 image data set consists of 586 MRI studies on 162 subjects in the UCSF image database. This is also the study cohort used as the Test Phase data in the QIN BMMR Challenge.
| Table 5. 45 subjects excluded from Level 2b set |
|---|
ID 1027 ID 1040 ID 1045 ID 1046 ID 1048 ID 1054 ID 1063 ID 1067 | ID 1079 ID 1084 ID 1103 ID 1110 ID 1120 ID 1137 ID 1139 ID 1152 | ID 1157 ID 1159 ID 1160 ID 1167 ID 1171 ID 1176 ID 1177 ID 1180 | ID 1182 ID 1185 ID 1187 ID 1189 ID 1192 ID 1194 ID 1203 ID 1206 | ID 1210 ID 1212 ID 1214 ID 1215 ID 1219 ID 1221 ID 1222 ID 1228 | ID 1234 ID 1235 ID 1237 ID 1238
ineligible: Case #: 128 |
- Hylton, N. M., Gatsonis, C. A., Rosen, M. A., Lehman, C. D., Newitt, D. C., Partridge, S. C., Bernreuter, W. K., Pisano, E. D., Morris, E. A., Weatherall, P. T., Polin, S. M., Newstead, G. M., Marques, H. S., Esserman, L. J., & Schnall, M. D. (2016). Neoadjuvant Chemotherapy for Breast Cancer: Functional Tumor Volume by MR Imaging Predicts Recurrence-free Survival—Results from the ACRIN 6657/CALGB 150007 I-SPY 1 TRIAL. https://doi.org/10.1148/radiol.2015150013 PMC4819899
|