Summary
This is a dataset with multiparametric prostate MRI applied in a test-retest setting, allowing to evaluate repeatability of the MRI-based measurements in the prostate. There is very limited data about the repeatability in mpMRI of the prostate, while such information is critical for establishing technical characteristics of mpMRI as imaging biomarker of prostate cancer. Data was provided by the Brigham and Women's Hospital team. Data collection was supported by U01 CA151261 (PI Fiona Fennessy). Preparation of data for public sharing was supported by U24 CA180918 (http://qiicr.org) (MPI Andrey Fedorov and Ron Kikinis). |
Type of cancer: Confirmed or suspected prostate cancer
Acquisition Protocol: Standard prostate mpMRI protocol implemented at Brigham and Women's Hospital was used in this study. For a given patient, we aimed to maintain similar protocol settings, and used the same scanner hardware and software configurations for both the baseline and repeat examinations, which were acquired within 2 weeks of time. All of the imaging studies were acquired at 3 Tesla magnet strength. Due to the scanner hardware upgrade in the middle of the study, 6 of the patients had baseline and repeat study performed on a GE Signa HDxt platform, software release 15.0_M4A_097.a, while the remaining 7 patients were scanned on a GE Discovery MR750w, software release DV24.0_R01_1344 (General Electric Healthcare, Milwaukee, WI). Transrectal coil within an air-filled balloon (Medrad Inc., Warrendale, PA) was used in all imaging studies. mpMRI protocol included T2-weighted, Diffusion Weighted (DW) (b-values of 0 and 1400 mm/s2) and Dynamic Contrast Enhanced (DCE) sequences. Detailed acquisition parameters are listed in Table 1 of [1]. DWI Apparent Diffusion Coefficient (ADC) and DCE subtract maps (further referred to as SUB; computed as the difference between the phase corresponding to the contrast bolus arrival and the baseline phase) were generated using the scanner software.
The imaging data is accompanied by the following types of derived data:
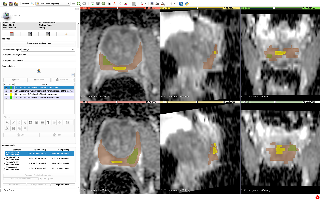
- manual segmentations of the total prostate gland, peripheral zone of the prostate gland, suspected tumor and normal regions (where applicable). Segmentations were done by a radiologist with the expertise in prostate MRI
- volume measurements (for axial T2w images and ADC images) and mean ADC (for ADC images) corresponding to the segmented regions.
Both segmentations and segmentation-based measurements are stored as DICOM objects (DICOM Segmentation images and DICOM Structured Reports that follow DICOM SR TID 1500). For the details about data representation and tools available to convert and visualize the data see [2].
In the future we plan to augment this dataset with the parametric maps obtained using that analysis (in DICOM), and potentially (pending IRB clearance) clinical data (demographics, PSA), pathology sampling data (biopsy Gleason score) and results of PI-RADS interpretation.
References:
[1] Fedorov A, Vangel MG, Tempany CM, Fennessy FM. Multiparametric Magnetic Resonance Imaging of the Prostate: Repeatability of Volume and Apparent Diffusion Coefficient Quantification. Investigative Radiology. 52, 538–546 (2017). DOI: 10.1097/RLI.0000000000000382
[2] Fedorov, A., Schwier, M., Clunie, D., Herz, C., Pieper, S., Kikinis,R., Tempany, C. & Fennessy, F. An annotated test-retest collection of prostate multiparametric MRI. Scientific Data 5, 180281 (2018). DOI: 10.1038/sdata.2018.281
About the NCI QIN
The mission of the QIN is to improve the role of quantitative imaging for clinical decision making in oncology by developing and validating data acquisition, analysis methods, and tools to tailor treatment for individual patients and predict or monitor the response to drug or radiation therapy. More information is available on the Quantitative Imaging Network Collections page. Interested investigators can apply to the QIN at: Quantitative Imaging for Evaluation of Responses to Cancer Therapies (U01) PAR-11-150.
|