Pre-clinical animal models of spontaneous metastatic cancer are infrequent; the few that exist are resource intensive because determination of the presence of metastatic disease, metastatic burden, and response to therapy normally require multiple timed cohorts with animal sacrifice and extensive pathological examination. We identified and characterized a patient derived xenograft model with metastatic potential, adenocarcinoma colon xenograft 997537-175-T. In this study we performed a detailed imaging characterization (workflow below) of this model, which develops spontaneous lung metastases, details are provided in the attached standard operating procedures. Tumors in half of the mice were resected in the range 200-300 mm3 size; tumors in the other half were allowed to grow until it was necessary to euthanize them because of tumor size. |
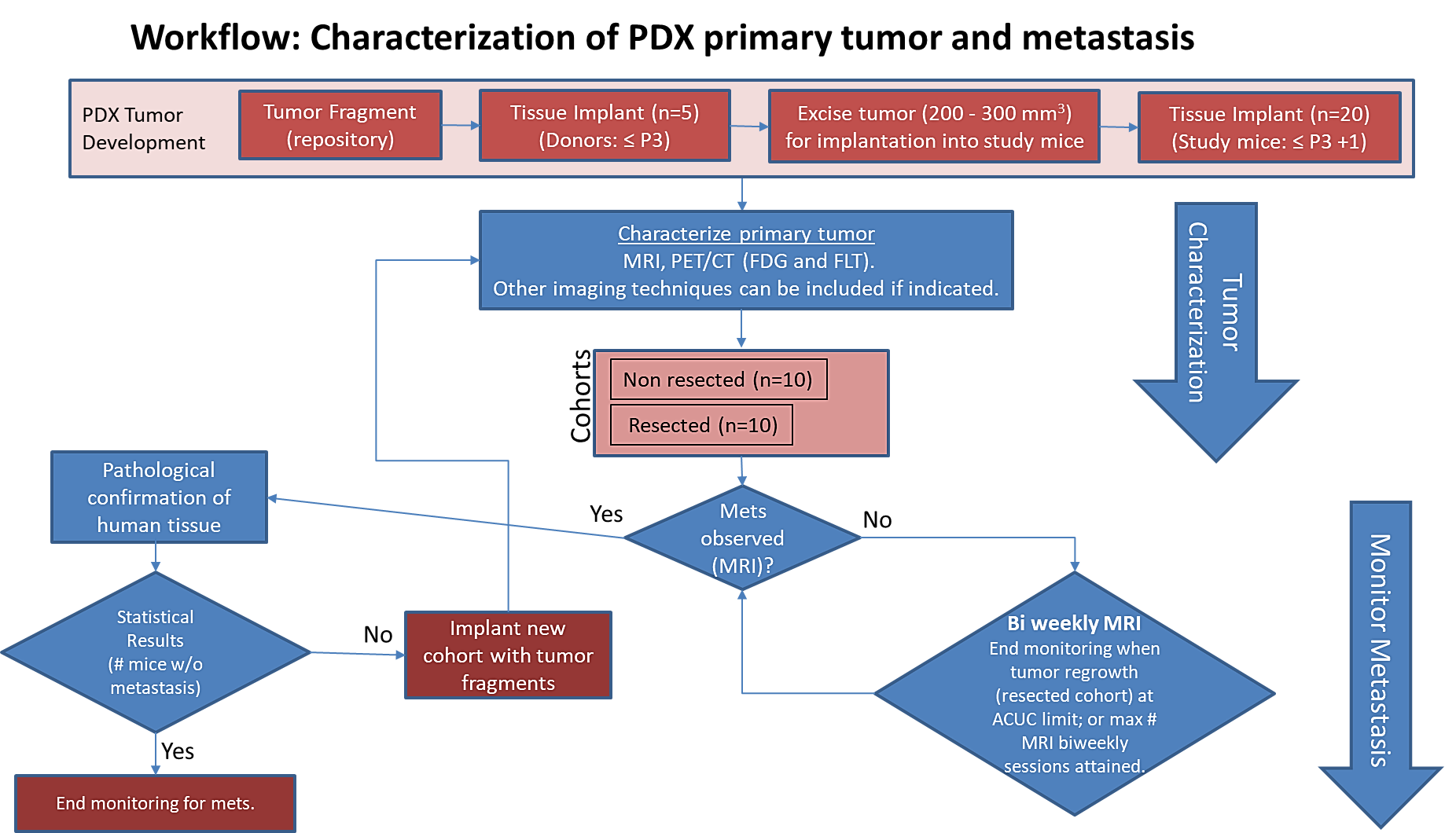
The imaging characteristics of this model (PDMR-997537-175-T), which is available from the National Cancer Institute Patient-Derived Models Repository (https://pdmr.cancer.gov/), is highly favorable for preclinical research studies of metastatic disease when used in conjunction with non-contrast T2 weighted MRI.
Table 1: Penetrance and location of pathological confirmed metastatic lesion(s).
# animals in group | # animals that displayed metastasis in MRI and confirmed by Pathology | Pathology confirmation of MRI (primary imaging site) | Other confirmed Location (s) | Mouse ID: MRI with pathology confirmation of metastasis |
10 (non-resected) | 6 (4 mice were EU due to xenograft size prior to observation of metastasis) | Lung | Lymph Node | 1319, 1321, 1326, 1327, 1334, 1338 |
10 (resected) | 10 | Lung | Lymph Node | 1320, 1323, 1324, 1325, 1328, 1329, 1332, 1333, 1335, 1337 |
Percent penetrance with respect to the average time-to-metastasis for non-resected (plot A: time from implant) and resected (plot B: time from tumor resection) cohorts.
 Plot A |
|
|---|
PET/CT Characterization of the primary tumor: Baseline PET (SOP attached) were performed when tumor reached an approximate 200 mm3. Average SUVmax values (n=5) were calculated; [18F]FDG: 4.4 ± 0.6 and [18F]FLT: 1.3 ± 0.3.
Excellent metastatic model with at least 60% penetrance non-resected and 100% with planned resection. Metastases well observed on T2 MRI imaging allowing non-invasive evaluation in treatment trials. This model can be challenging in a drug study due to the rapid xenograft tumor growth and re-growth after tumor excision.
We would like to acknowledge the individuals and institutions that have provided data for this collection:
|