Versions Compared
Key
- This line was added.
- This line was removed.
- Formatting was changed.
Summary
| Excerpt |
|---|
| ACRIN 6657 was designed as a prospective study to test MRI for ability to predict response to treatment and risk-of-recurrence in patients with stage 2 or 3 breast cancer receiving neoadjuvant chemotherapy (NACT). ACRIN 6657 was conducted as a companion study to CALGB 150007, a correlative science study evaluating tissue-based biomarkers in the setting of neoadjuvant treatment of breast cancer. Collectively, CALGB 150007 and ACRIN 6657 formed the basis of the multicenter Investigation of Serial Studies to Predict Your Therapeutic Response with Imaging and moLecular Analysis (I-SPY TRIAL) breast cancer trial, a study of imaging and tissue-based biomarkers for predicting pathologic complete response (pCR) and recurrence-free survival (RFS). |
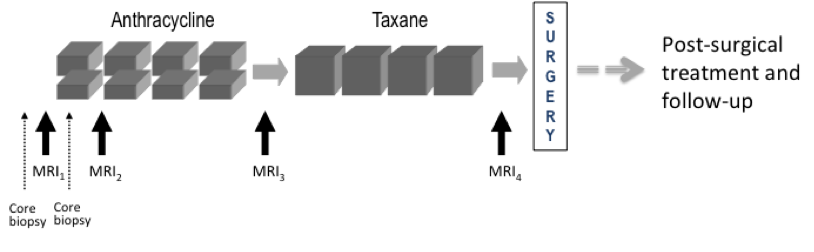
Participant Eligibility and Enrollment: Criteria for inclusion were patients enrolling on CALGB 150007 with T3 tumors measuring at least 3 cm in diameter by clinical exam or imaging and receiving neoadjuvant chemotherapy with an anthracycline-cyclophosphamide regimen alone or followed by a taxane. Pregnant patients and those with ferromagnetic prostheses were excluded from the study. The study was open to enrollment from May 2002 to March 2006. 237 patients were enrolled, of which 230 met eligibility criteria.
Acknowledgements
This shared data set was provided by David Newitt, PhD and Nola Hylton, PhD from the Breast Imaging Research Program at UCSF, in collaboration with ACRIN, CALGB, the I-SPY TRIAL, and TCIA. Many thanks are due to The ACRIN 6657 trial team, The I-SPY 1 TRIAL team, and all the patients participating in these studies
Funding sources include NIH grants to UCSF (R01 CA132870 and U01 CA151235), ACRIN (UO1 CA079778 and UO1 CA080098), and CALGB (UO1 CA31964 and UO1 CA33601).
| Localtab Group | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HTML |
|---|
<script type='text/javascript' src='https://d1bxh8uas1mnw7.cloudfront.net/assets/embed.js'></script>
<div data-badge-popover="right" data-badge-type='donut' data-doi="10.7937/K9/TCIA.2016.HdHpgJLK" class="altmetric-embed"></div>
<meta name="DC.Identifier" content="10.7937/K9/TCIA.2016.HdHpgJLK" />
<meta name="citation_doi" content="10.7937/K9/TCIA.2016.HdHpgJLK" />
<meta name="citation_title" content=" Multi-center breast DCE-MRI data and segmentations from patients in the I-SPY 1/ACRIN 6657 trials." /> |
Summary
| Excerpt |
|---|
| ACRIN 6657 was designed as a prospective study to test MRI for ability to predict response to treatment and risk-of-recurrence in patients with stage 2 or 3 breast cancer receiving neoadjuvant chemotherapy (NACT). ACRIN 6657 was conducted as a companion study to CALGB 150007, a correlative science study evaluating tissue-based biomarkers in the setting of neoadjuvant treatment of breast cancer. Collectively, CALGB 150007 and ACRIN 6657 formed the basis of the multicenter Investigation of Serial Studies to Predict Your Therapeutic Response with Imaging and moLecular Analysis (I-SPY TRIAL) breast cancer trial, a study of imaging and tissue-based biomarkers for predicting pathologic complete response (pCR) and recurrence-free survival (RFS). |
Participant Eligibility and Enrollment: Criteria for inclusion were patients enrolling on CALGB 150007 with T3 tumors measuring at least 3 cm in diameter by clinical exam or imaging and receiving neoadjuvant chemotherapy with an anthracycline-cyclophosphamide regimen alone or followed by a taxane. Pregnant patients and those with ferromagnetic prostheses were excluded from the study. The study was open to enrollment from May 2002 to March 2006. 237 patients were enrolled, of which 230 met eligibility criteria.
Acknowledgements
This shared data set was provided by David Newitt, PhD and Nola Hylton, PhD from the Breast Imaging Research Program at UCSF, in collaboration with ACRIN, CALGB, the I-SPY TRIAL, and TCIA. Many thanks are due to The ACRIN 6657 trial team, The I-SPY 1 TRIAL team, and all the patients participating in these studies
Funding sources include NIH grants to UCSF (R01 CA132870 and U01 CA151235), ACRIN (UO1 CA079778 and UO1 CA080098), and CALGB (UO1 CA31964 and UO1 CA33601).
| Localtab Group | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Level 2a: Good SER Volume Dataset – updated 9/3/14, 9/17/16This data set is comprised of the patient studies which, following quality reviews in 2014 and 2016, were judged to have sufficiently good image quality and protocol compliance for volumetric DCE SER analysis. Rejection criteria included: incomplete volumetric DCE acquisitions, lack of a 2nd post-contrast acquisition, variability in fat suppression across the image, observed patient motion during the DCE acquisition, significant DCE protocol deviations such as changing scan parameters or image position during DCE acquisition. Level 2a image data set consists of 706 MR studies on 207 subjects in the UCSF image database. These include 7 studies not included in Level 1 (no MRI LD recorded) as listed in Table 2. Table 2. Studies in Level 2a (good volumetric analysis) that do NOT have LD measures: ID 1059, T4 ID 1192, T2 ID 1215, T1
Level 2b: SER Volume Dataset Reported in Hylton et al. (Radiology, 2012) *This data set is comprised of the patient studies analyzed for pCR outcome and reported in the 2012 Radiology paper on ACRIN 6657 pCR results *. This data set is not provided as a shared list, as it is not recommended for use in further analysis. It is described here because it is the data set from which the Level 3 (primary aim analysis) set was derived. Inclusion and exclusion was determined by quality and protocol reviews available at that time. In addition to the exclusion criteria listed for Level 2a, studies done with imaging in the axial plane, in violation of the sagittal orientation specified in the trial imaging protocol, were excluded due to processing limitations of the analysis software. Similarly, bi-lateral sagittal acquisitions (alternating left and right volumetric acquisitions) were excluded. Level 2b image data set consists of 707 MRI studies on 207 subjects in the UCSF image database.
Tables 3 and 4 show the specific inclusion/exclusion differences between Levels 2a and 2b:
Table 4. 17 studies rejected since 2008 (in Level 2b but not in 2a) | Study ID and TP | Reason for rejection for volumetric SER analysis (level 2a) | 1007 T4 * No fatSat; Different protocol from T1 | 1035 T4 * | Only 1 post scan then acq. parameters changed | 1045 T1 | Alternating laterality acquisitions, 2 minute time gap | 1047 T1 * | Image position changed during DCE | 1053 T2 * | Alternating laterality acquisitions, bad pre- acquisition | 1053 T4 * | Alternating laterality acquisitions | 1055 T1 * | Alternating laterality acquisitions, 4 minute time gap | 1086 T1 * | Alternating laterality acquisitions, time gap, different protocol from T4 | 1091 T1 * | Changing acq. parameters during DCE | 1095 T2 * | Only 1 post scan then acq. parameters changed | 1173 T3 * | Off protocol timing | 1206 T1 | Bad DCE timing, 20 minute delay | 1206 T2 | Bad DCE timing, 1'29" acquisition time | 1224 T3 * | Scan position changed during DCE | 1230 T3 * | Scan position changed during DCE | #128 T1, T2 | 2 studies for ineligible patient: ACRIN ID 128 (no I-SPY ID) |
Level 3: Subset of Level 2b used in primary aim analysis, reported in Hylton et al. (Radiology, 2016) *This data set is comprised of the patient studies analyzed for RFS outcome and reported in the 2015 Radiology paper on ACRIN 6657 survival results (Hylton et al, Radiology *). Table 5 shows the 45 patients excluded from the level 2a cohort for this analysis. Please see the publication for specific information on exclusions of patients from this group. Level 3 image data set consists of 586 MRI studies on 162 subjects in the UCSF image database. This is also the study cohort used as the Test Phase data in the QIN BMMR Challenge. Table 5. 45 subjects excluded from Level 2b set | ID 1027 ID 1040 ID 1045 ID 1046 ID 1048 ID 1054 ID 1063 ID 1067 ID 1079 ID 1084 ID 1103 ID 1110 ID 1120 ID 1137 ID 1139 ID 1152 ID 1157 ID 1159 ID 1160 ID 1167 ID 1171 ID 1176 ID 1177 ID 1180 ID 1182 ID 1185 ID 1187 ID 1189 ID 1192 ID 1194 ID 1203 ID 1206 ID 1210 ID 1212 ID 1214 ID 1215 ID 1219 ID 1221 ID 1222 ID 1228 ID 1234 ID 1235 ID 1237 ID 1238 ineligible: Case #: 128
Localtab |
Other Publications Using This DataTCIA maintains a list of publications that leverage our data. At this time we are not aware of any publications based on this data. If you have a publication you'd like to add please contact the TCIA Helpdesk. Altmetrics
Citations & Data Usage PolicyThis collection is freely available to browse, download, and use for commercial, scientific and educational purposes as outlined in the Creative Commons Attribution 3.0 Unported License. See TCIA's Data Usage Policies and Restrictions for additional details. Questions may be directed to help@cancerimagingarchive.net. Please be sure to include the following citations in your work if you use this data set: like to add please contact the TCIA Helpdesk.
Other Publications Using This DataTCIA maintains a list of publications that leverage our data. At this time we are not aware of any publications based on this data. If you have a publication you'd
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||