Summary
| Excerpt |
|---|
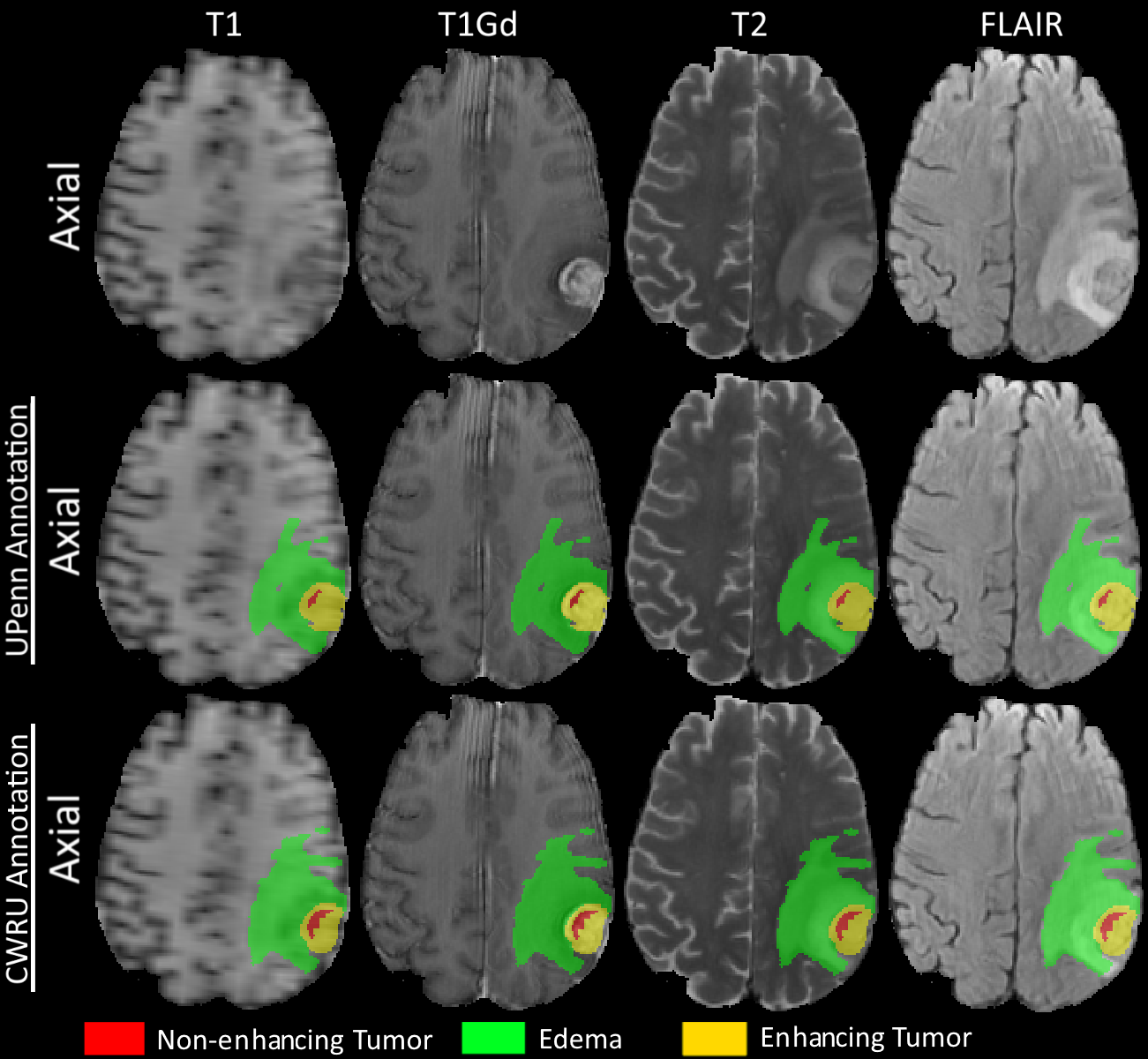
This dataset comprises oftwo paired sets of manually-correctedexpert segmentation labels for tumor sub-compartments of the pre-operative multi-institutional scans of the Glioblastom IvyGAPcollection of The Cancer Imaging Archive (TCIA). The paired sets of manually correctedThese labels have been approved by independent expert board-certified neuroradiologists at the Hospital of the University of Pennsylvania and at Case Western Reserve University. Furthermore, for each of the paired sets of approved labels, a diverse comprehensive panel of radiomic features is provided, along with their corresponding skull-stripped and co-registered multi-parametric magnetic resonance imaging (mpMRI) volumes (i.e. native (T1) and post- precontrast ,T1-weighted (T1- post contrastGd), T2, T2-FLAIR) magnetic resonance imaging (mpMRI) volumes, in NIfTI format. PreThe pre-operative mpMRI scans were identified in the Ivy GAP collection via radiological assessment. These scans were initially skull-stripped and co-registered to a common anatomical atlas (provided within this dataset), before their tumor segmentation labels were produced by different approachesfollowing a consistent annotation protocol across the two institutions , but consistent within each one. The segmentations were then revised, and any label misclassifications were manually corrected by an expert board-certified neuroradiologist. Thefinal labels were used to extract a rich panel of imaging features, includingradiomic features through the Cancer Imaging Phenomics Toolkit (CaPTk), comprising intensity, volumetric, morphologic, histogram-based, and textural parameters (including the novel COLLAGE featurescompliant with the Image Biomarker Standardisation Initiative (IBSI), as well as spatial information and diffusion properties extracted from glioma growth modelsthrough a 3D Slicer extension for the novel COLLAGE feature family. Radiomic features robust to variability in segmentations were then identified following a statistical robustness analysis. The approved expert segmentation labels should enable quantitative computational and clinical studies without the need to repeat manual annotations, whilst allowing for comparison across studies. They can also serve as a set of manually-annotated gold standard labels for performance evaluation in computational competitions, such as the International Brain Tumor Segmentation (BraTS) challenge. The provided panel of robust radiomic features may facilitate research integrative of the molecular characterization offered by the Allen Institute, and hence allow associations with molecular markers (radiogenomics), clinical outcomes, treatment responses and other endpoints, by researchers without sufficient computational background to extract such features . |
Acknowledgements
We would like to acknowledge the individuals and institutions that have provided data for this collection:
Spyridon Bakas, Ph.D., Center for Biomedical Image Computing & Analytics, Perelman School of Medicine, University of Pennsylvania, Philadelphia, PA, USA.
...
. The complete reproducibility analysis can be found in the associated publication citation found in the “Citations & Data Usage Policy”. Specifically, the released data comprises of 1) the available expert segmentation labels of the various tumor sub-compartments performed at each institution (i.e. 34 subjects segmented at UPenn, 34 subjects segmented at CWRU), with a total of 37 subjects (including 31 paired segmentations performed at both UPenn and CWRU), in the original space they were created (i.e., SRI for UPenn and MNI for CWRU), with 2) their corresponding co-registered and skull-stripped structural mpMRI scans (i.e., in SRI for UPenn and in MNI for CWRU), 3) the paired expert segmentation labels that were available for the 31 subjects, all being co-registered in the SRI atlas, 4) the corresponding SRI and MNI anatomical atlas files that we employed, 5) the complete set of 11,700 extracted radiomic features per subject, for each of the 31 included subjects, 6) the metadata relating to the metrics we utilized for the evaluation of the inter-rater agreement, as well as 7) the parameters used for the radiomic feature extraction and the correlation analysis results for identifying robust radiomic features, for the 28 subjects, and finally 8) the specific identified robust/reproducible radiomic features. All image related files are provided in NIfTI format, while the metadata files are provided in tabular formats (.xlsx and .csv). MNI atlas: see (Montreal Neurological Institute, https://mcin.ca/research/neuroimaging-methods/atlases/ ) SRI atlas: see (T. Rohlfing, et al. (2010) DOI: 10.1002/hbm.20906 , PMC2915788) AcknowledgementsThe authors would like to acknowledge the following funding sources:
The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH, U.S. Department of Veterans Affairs, the DoD, or the United States Government. |
| Localtab Group | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|