Versions Compared
Key
- This line was added.
- This line was removed.
- Formatting was changed.
Summary
In 2004 when presenting the NCI Executive Committee the ACRIN proposal to conduct the CTC trial, a case was made that publicly accessible image data sharing would offer a valuable research asset to a wide image processing research community. Adding to the many merits of that proposal, the data-sharing component was strongly endorsed. ACRIN completed the trial expeditiously and its results were published in NEJM in fall 2008 to wide interest. ACRIN has graciously allowed the wider research community access to a portion of the data from that trial here on TCIA, including spreadsheets identifying positive and negative polyp cases. The complete ACRIN protocol description can be found atThe National Institutes of Health Clinical Center performed 82 abdominal contrast enhanced 3D CT scans (~70 seconds after intravenous contrast injection in portal-venous) from 53 male and 27 female subjects. Seventeen of the subjects are healthy kidney donors scanned prior to nephrectomy. The remaining 65 patients were selected by a radiologist from patients who neither had major abdominal pathologies nor pancreatic cancer lesions. Subjects' ages range from 18 to 76 years with a mean age of 46.8 ± 16.7. The CT scans have resolutions of 512x512 pixels with varying pixel sizes and slice thickness between 1.5 − 2.5 mm, acquired on Philips and Siemens MDCT scanners (120 kVp tube voltage).
A medical student manually performed slice-by-slice segmentations of the pancreas as ground-truth and these were verified/modified by an experienced radiologist.
Data Example
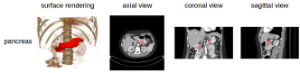 Image Added
Image Added
Note
The DICOM files were created from anonymized volumetric images (Analyze and NifTI) using this from ITK: http://www.
acrinTabID151/Default.aspxhtml/Examples_2IO_2ImageReadDicomSeriesWrite_8cxx-example.html.
| Localtab Group | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|