Summary
| Excerpt |
|---|
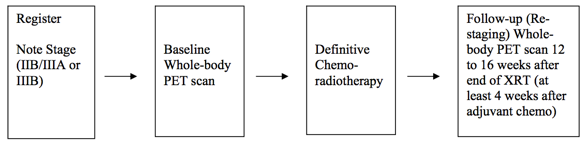
Positron Emission Tomography Pre- and Post-treatment Assessment for Locally Advanced Non-small Cell Lung Carcinoma This was a multicenter clinical trial by the ACRIN Cooperative Group (now part of ECOG-ACRIN) and the RTOG Cooperative Group (now part of NRG) using FDG-PET imaging both pre- and post-chemoradiotherapy. The objective of the ACRIN 6668 multi-center clinical trial was to determine if the PET standardized uptake value (SUV) measurement The primary purpose of this study is to determine if the SUV measurement from FDG-PET imaging shortly after treatment is a useful predictor of predictor of long-term clinical outcome (survival) after definitive chemoradiotherapy. Eligible patients are were those older than 18 years with AJCC clinical stage -criteria clinical stage IIB/III nonIII non-small cell lung carcinoma who are were being planned for definitive concurrent definitive concurrent chemoradiotherapy (inoperable disease). Primary Aim Findings: Higher post-treatment tumor SUV (SUVpeak, SUVmax) is associated with worse survival in stage III NSCLC, although a clear SUV cutoff value for routine clinical use as a prognostic factor was uncertain [1]. Later analyses found that larger pre-treatment metabolic tumor volumes (MTVs) were associated with significantly worse overall survival [2]. Other secondary analyses found potentially predictive image texture biomarkers. Study Design Summary: Patients received conventional concurrent platinum-based chemoradiotherapy without surgery; post-radiotherapy consolidation chemotherapy was allowedA total of 250 participants will be enrolled, including at least 75 with stage IIB/IIIA and at least 75 with stage IIIB disease. A baseline whole-body FDGbody FDG-PET scan will be was performed . Follow up after chemoradiotherapy will comprise restaging of the disease and a second prior to therapy. A second post-treatment whole-body FDG-PET scan 12 to 16 weeks after the completion of occurred approximately 14 weeks after radiotherapy (at least 4 weeks after adjuvant chemotherapy). Pre-treatment FDG-PET scans were performed on ACRIN-qualified scanners. Post-treatment FDG-PET scans were required to be performed within 12–16 weeks after completion of therapy, using the same scanner as that used for the pre-treatment scans. Acknowledgements This shared data set was provided by <<<others>>>> in collaboration with the with the American College of Radiology Imaging network (ACRIN). Many Core Lab. Many thanks are due to the to the ACRIN 6668 trial team, and and all the patients participating in the study. This study was supported by ACRIN, the ACRIN Cooperative Group (now part of ECOG-ACRIN) and the RTOG Cooperative Group (now part of NRG) which received funding from the National Cancer Institute through <<<>>>, under the American Recovery and Reinvestment ACT of 2009 (ARRA) and UO1 <<<>>>UO1 CA080098 and UO1 CA079778. |
| Localtab Group | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
...