Summary
| Excerpt |
|---|
 Image Removed Image RemovedPre-clinical animal models of spontaneous metastatic cancer are infrequent; the few that exist are resource intensive because determination of the presence of metastatic disease, metastatic burden, and response to therapy normally require multiple timed cohorts with animal sacrifice and extensive pathological examination. We identified and characterized a patient derived xenograft model with metastatic potential, melanoma xenograft 425362-245-T. In this study we performed a detailed imaging characterization (workflow below) of this model, which develops spontaneous lung metastases, details are provided in the attached standard operating procedures. Tumors in half of the mice were resected in the range 200-300 cm3 size; tumors in the other half were allowed to grow until it was necessary to euthanize them because of tumor size. |
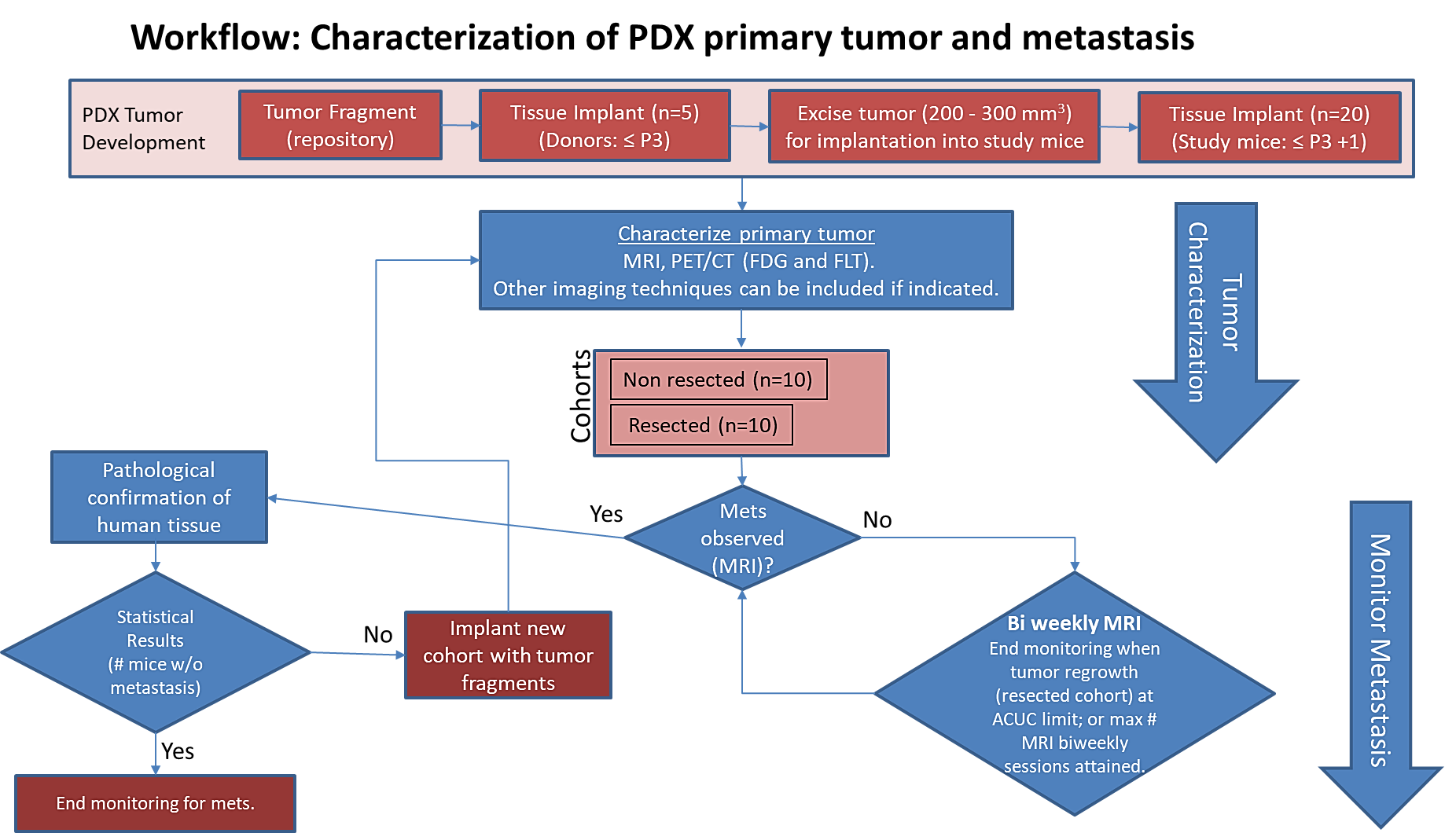 Image Added
Image Added
The imaging characteristics of this model
, (PDMR-425362-245-T) which is available from the National Cancer Institute Patient-Derived Models Repository (https://pdmr.cancer.gov/), is highly favorable for preclinical research studies of metastatic disease when used in conjunction with non-contrast T2 weighted MRI.
Table 1: Penetrance and location of pathological confirmed metastatic lesion(s).
# animals in group | # animals that displayed metastasis in MRI and confirmed by Pathology | Pathology confirmation of MRI (primary imaging site) | Other confirmed Location (s) | Mouse ID: MRI with pathology confirmation of metastasis |
10 (non-resected)
| 4 (6 mice were EU due to xenograft size prior to observation of metastasis) |
Lung |
Kidney |
1512, 1516, 1518, 1520 |
10 (resected) | 10 | Lung | Kidney, Liver, Pancreas
| 1506, 1508, 1509, 1510, 1511, 1515, 1517, 1519, 1521, 1523 |
Percent penetrance with respect to the average time-to-metastasis for non-resected (plot A: time from implant) and resected (plot B: time from tumor resection) cohorts.
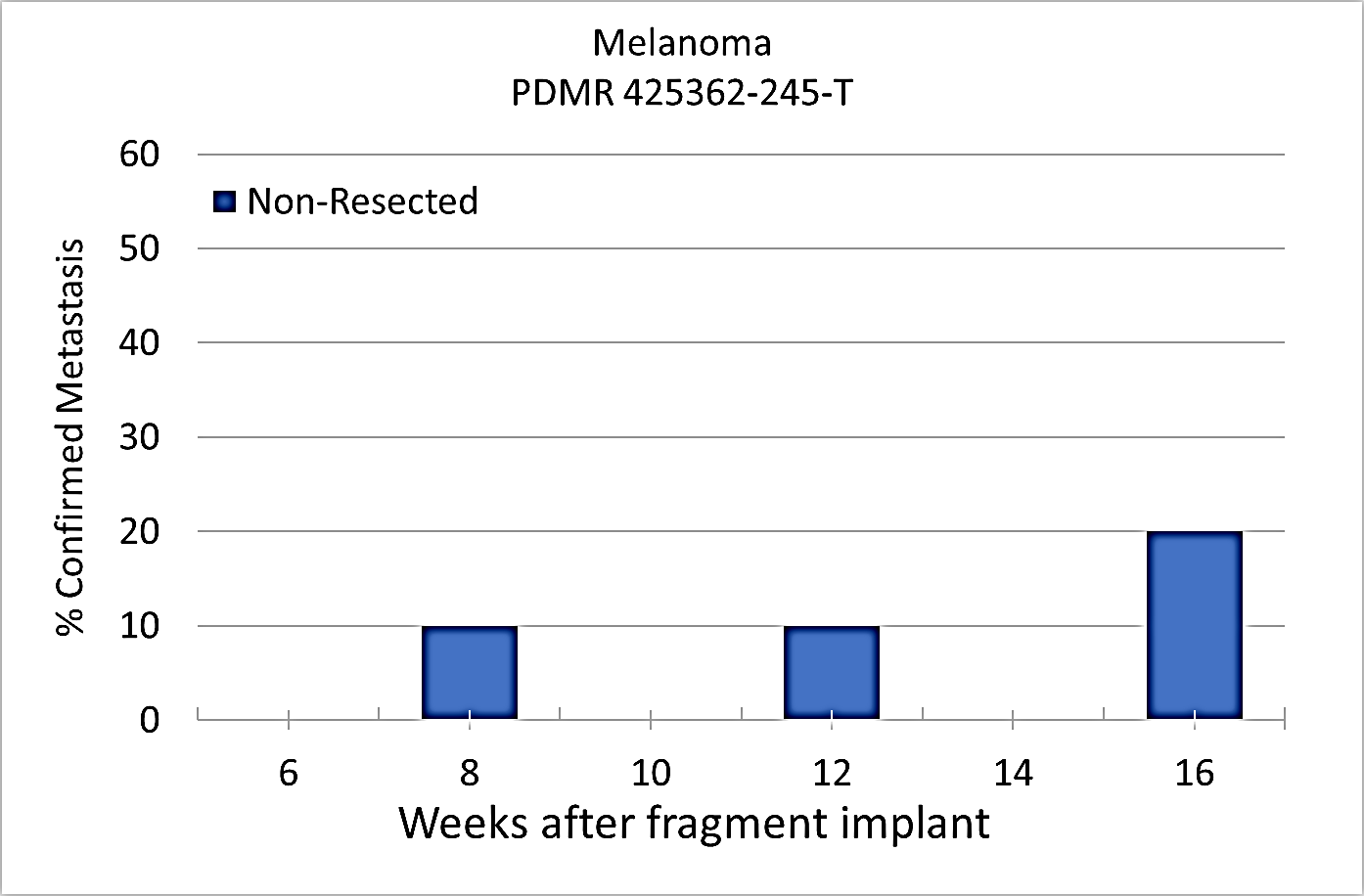 Image Added Image Added
Plot A | 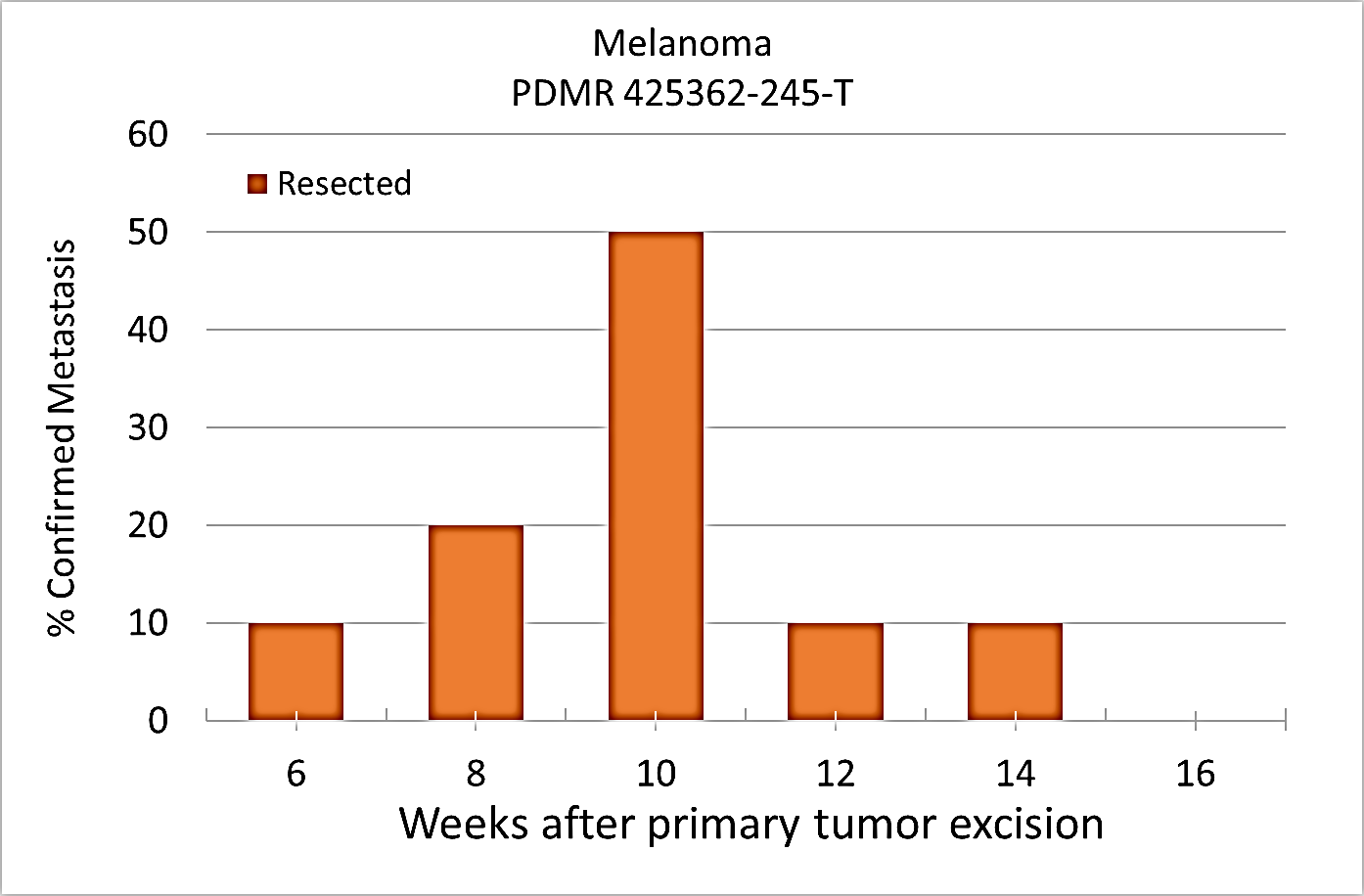 Image Added Image Added
Plot B |
|---|
PET/CT Characterization of the primary tumor: Baseline PET (SOP attached) were performed when tumor reached an approximate 200 mm3. Average SUVmax values (n=5) were calculated; [18F]FDG: 2.7 ± 0.5 and [18F]FLT: 2.0 ± 0.3.
Conclusion:
Melanoma PDMR-425362-245-T model can be challenging due to the rapid growth of the xenograft and regrowth. Metastases was well observed on T2 MRI imaging allowing non-invasive evaluation in treatment trials.
Acknowledgements
We would like to acknowledge the individuals and institutions that have provided data for this collection:
- Frederick National Laboratory for Cancer Research – Special Thanks to Joseph D. Kalen, PhD, Lilia V. Ileva, MS, Lisa A Riffle, Nimit Patel, Keita Saito, PhD, Yvonne Evrard, PhD, Elijah Edmondson, DVM, PhD, Jessica Phillips, Simone Difilippantonio, PhD, Chelsea Sanders, Amy JanesJames, Lia Thang, Ulrike Wagner, Yanling Liu, PhD, John B. Freymann, and Justin Kirby.
- Division of Cancer Therapeutics and Diagnosis/National Cancer Institute - James L. Tatum, MD, Paula M Jacobs, PhD, Melinda G. Hollingshead, DVM, and James H. Doroshow, MD
- PixelMed Publishing – Special Thanks to David A. Clunie, MD
- University of Arkansas for Medical Sciences – Special Thanks to Kirk E. Smith
- This project has been funded in whole or in part with Federal funds from the National Cancer Institute, National Institutes of Health, under Contract No. HHSN261201500003I. The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. Government.
| Localtab Group |
|---|
| Localtab |
|---|
| active | true |
|---|
| title | Data Access |
|---|
| Click the Download button to save a ".tcia" manifest file to your computer, which you must open with the NBIA Data Retriever . Click the Search button to open our Data Portal, where you can browse the data collection and/or download a subset of its contents.
| Data Type | Download all or Query/Filter |
|---|
| Images (DICOM, 2.3 GB) | | | Supplemental Data (format) |  Image Removed Image Removed
| (Download requires NBIA Data Retriever) | | Tracking spreadsheet for PDMR-425362-245-T | | Tcia button generator |
|---|
| url | https://wiki.cancerimagingarchive.net/download/attachments/70223880/SAIP%20PDM%20Overview_TCIA.xlsx?api=v2 |
|---|
| |
| | SR Conversion Routine (Converts a tracking spreadsheet into a DICOM Structured Report) | | Tcia button generator |
|---|
| url | https://wiki.cancerimagingarchive.net/download/attachments/70223880/MakeDICOMSRFromSAIPPDMOverview_SourceCode_20200831.zip?api=v2 |
|---|
| |
| | PDMR Patient Specimen (external) | | | Standard Operating Procedure 50101 MRI T2 Weighted Non-Contrast Protocol Single Mouse Pulmonary Gated and Multi-Mouse Non-Gated | | | Standard Operating Procedure 50102 Positron Emission Tomography (PET) imaging protocol | |
Click the Versions tab for more info about data releases.
|
| Localtab |
|---|
| title | Detailed Description |
|---|
|
| |
|---|
Modalities | MR, SR | Number of Subjects | 20 | Number of Studies | 115 | Number of Series | 210 | Number of Images | 3509 | | Images Size (GB) | 2.3 GB | # animals in group# animals that displayed metastasis in MRI and confirmed by Pathology | Pathology confirmation of MRI (primary imaging site) | Other confirmed Location (s) | 10 (non-resected) 6 (4 mice were EU due to xenograft size prior to observation of metastasis) | Lung | Lymph node | 10 (resected) | 10 | Lung | Lymph node |  Image Removed Image Removed Image Removed Image Removed
Baseline PET [18F]FDG SUVmax: 4.4 ± 0.6 Baseline PET [18F]FLT SUVmax: 1.3 ± 0.3
|
| Localtab |
|---|
| title | Citations & Data Usage Policy |
|---|
| | These collections are freely available to browse, download, and use for commercial, scientific and educational purposes as outlined in the Creative Commons Attribution 3.0 Unported License. Questions may be directed to help@cancerimagingarchive.net. Please be sure to acknowledge both this data set and TCIA in publications by including the following citations in your work:
| Tcia license 4 international |
|---|
| Info |
|---|
| Tatum, J. L., (https://orcid.org/0000-0002-3217-2478) Kalen, J. D., (https://orcid.org/0000-0002-7163-4604) Jacobs, P. M., (https://orcid.org/0000-0002-9423-6473) Ileva, L. V., (https://orcid.org/0000-0001-8286-8396) Riffle, L. A., (https://orcid.org/0000-0003-3975-3088) Keita, S., Patel, N., (https://orcid.org/0000-0002-8251-7155) Sanders, C., (https://orcid.org/0000-0001-8042-4783) James, A., Difilippantonio, S., (https://orcid.org/0000-0002-8234-1559) Thang, L., Hollingshead, M. G., (https://orcid.org/0000-0002-1207-1397) Phillips, J., Edmondson, E https://orcid.org/0000-0002-6106-3705 ., Evrard, Y., Clunie, D. A, (https://orcid.org/0000-0002-2406-1145) ., Liu, Y., Smith, K. E, (https://orcid.org/0000-0002-8735-7576) ., Wagner, U., (https://orcid.org/0000-0002-3230-5058) Freymann, J. B. Kirby, J., … Doroshow, J. H. (2020). Imaging characterization of a metastatic patient derived model of melanoma: PDMR-425362-245-T [Data set]. The Cancer Imaging Archive. https://orciddoi.org/0000-0003-3487-892210.7937/TCIA.2020.7YRS-7J97) Doroshow, J. H, (https://orcid.org/0000-0002-4463-1790)
|
| Info |
|---|
| This project has been funded in whole or in part with Federal funds from the National Cancer Institute, National Institutes of Health, under Contract No. HHSN261201500003I. The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. Government. |
| Info |
|---|
| Clark K, Vendt B, Smith K, Freymann J, Kirby J, Koppel P, Moore S, Phillips S, Maffitt D, Pringle M, Tarbox L, Prior F. The Cancer Imaging Archive (TCIA): Maintaining and Operating a Public Information Repository, Journal of Digital Imaging, Volume 26, Number 6, December, 2013, pp 1045-1057. DOI: https://doi.org/10.1007/s10278-013-9622-7 |
TCIA maintains a list of publications which leverage TCIA data. If you have a manuscript you'd like to add please contact the TCIA Helpdesk. |
| Localtab |
|---|
| 1 yyyymmdd | Data Type | Download all or Query/Filter |
|---|
| Images (DICOM, 2.3 GB) | | Requires )| Clinical Data (CSV) | Link | | Other (format) |  Image Removed Image Removed
| |
|
Type your task here, using "@" to assign to a user and "//" to select a due date| cancerimagingarchive.net/download/attachments/70223880/SAIP%20PDM%20Overview_TCIA.xlsx?api=v2 |
| |
| | SR Conversion Routine (Converts a tracking spreadsheet into a DICOM Structured Report) | | Tcia button generator |
|---|
| url | https://wiki.cancerimagingarchive.net/download/attachments/70223880/MakeDICOMSRFromSAIPPDMOverview_SourceCode_20200831.zip?api=v2 |
|---|
| |
| | PDMR Patient Specimen (external) | | | Standard Operating Procedure 50101 MRI T2 Weighted Non-Contrast Protocol Single Mouse Pulmonary Gated and Multi-Mouse Non-Gated | | | Standard Operating Procedure 50102 Positron Emission Tomography (PET) imaging protocol | |
Added two supplementary files: 1) an example tracking spreadsheet for multi-cohort murine studies and 2) an SR Conversion Routine to convert the tracking spreadsheet parameters into a DICOM Structured Report (SR). | Data Type | Download all or Query/Filter |
|---|
| Images (DICOM, 2.3 GB) | | | PDMR Patient Specimen (external) | | | Standard Operating Procedure 50101 MRI T2 Weighted Non-Contrast Protocol Single Mouse Pulmonary Gated and Multi-Mouse Non-Gated | | | Standard Operating Procedure 50102 Positron Emission Tomography (PET) imaging protocol | |
|
|