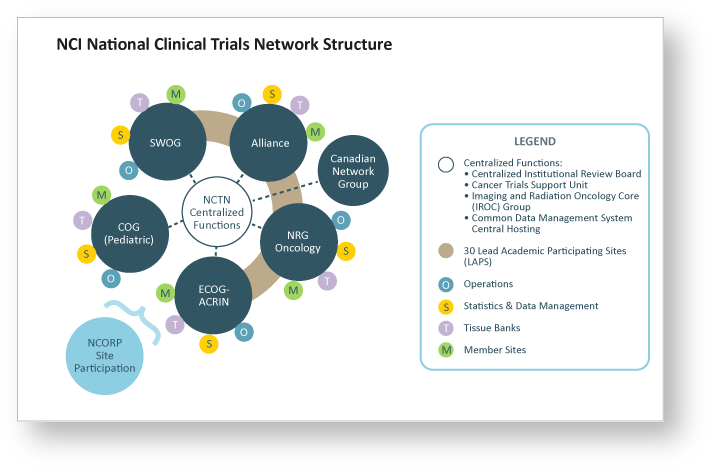
Imaging data from clinical trials provides the opportunity to link imaging characteristics to clinical trial analyses, associated clinical data and patient outcomes. From 2018 to 2023, the NCI Cancer Imaging Program Informatics Lab supported an additional TCIA image data collection center focused specifically on clinical trial data. Many clinical trial data sets originate from the NCI National Clinical Trials Network (NCTN), which is a collection of organizations and clinicians that coordinates and supports cancer clinical trials at more than 3,000 sites across the United States and Canada. The NCTN provides the infrastructure for NCI-funded treatment, screening, and diagnosis trials to improve the lives of patients with cancer. The NCTN explicitly requires data sharing of the patient-level clinical data in https://nctn-data-archive.nci.nih.gov/view-trials, and TCIA serves as the imaging repository for that archive.
Labeled data for tumor detection & segmentation
With the large increase in availability of such publicly available, de-identified datasets there is now an opportunity to substantially increase their value to the cancer imaging research community by curating, segmenting and labeling them to expedite progress in data driven fields such as radiomics and artificial intelligence. Recognizing this fact, NCI has provided funds to retrospectively generate 3d segmentations and seed points to identify tumor locations in additional trial datasets. The resulting data will be made freely available on TCIA, along with supporting documentation and sample code (e.g. Jupyter Notebooks) that will facilitate streamlined analysis on widely used data science platforms. This resource is being developed in a manner that will reduce the barriers for non-radiology researchers to use the data to develop predictive radiomic tools, to help developers of quantitative imaging software and machine learning models for tumor segmentation, detection and total burden estimations to assess response assessment.
Clinical Trial Datasets on TCIA