Summary
This collection contains clinical data and computed tomography (CT) from 22 non-small cell lung cancer (NSCLC) radiotherapy patients. For 21 of these patients with pre-treatment CT scans, repeated blinded manual delineations by five different radiation oncologists of the 3D volume of the gross tumor volume on CT and clinical outcome data are available. The above was repeated with the same set of five radiation oncologists, using an in-house autosegmentation tool for initial delineation followed by manual adjustment of the primary gross tumor volume outline. For one patient, clinical data and CT was available but the tumor delineations were not extracted. This patient was included in this collection for the sake of completeness.
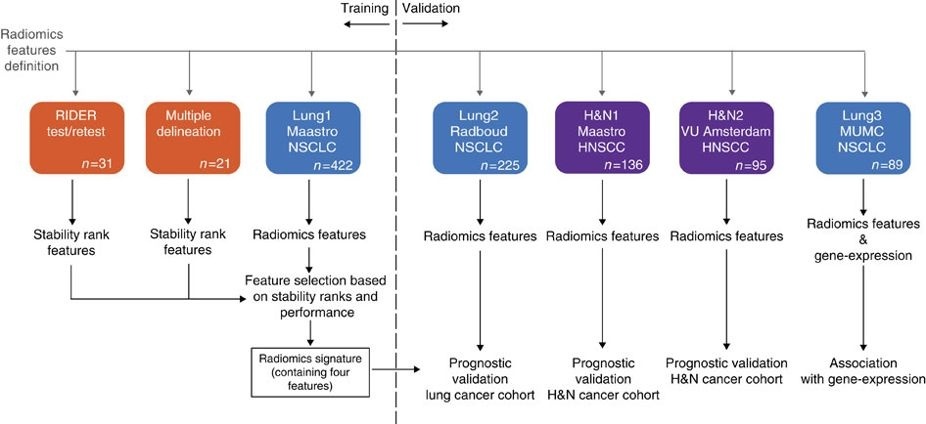
This dataset refers to the "Multiple delineation" dataset of the study published in Nature Communications (https://doi.org/10.1038/ncomms5006). In short, the publication used a radiomics approach to computed tomography data of 1,019 patients with lung or head-and-neck cancer. Radiomics refers to the comprehensive quantification of tumor phenotypes by applying a large number of quantitative image features. In the published analysis, 440 features quantifying tumor image intensity, shape, and texture were extracted. We found that a large number of radiomic features have prognostic power in independent data sets, many of which were not identified as significant before. Radiogenomics analysis revealed that a prognostic radiomic signature, capturing intra-tumor heterogeneity, was associated with underlying gene-expression patterns. These data suggest that radiomics identifies a general prognostic phenotype existing in both lung and head-and-neck cancer. This may have a clinical impact as imaging is routinely used in clinical practice, providing an unprecedented opportunity to improve decision-support in cancer treatment at low cost.
The delineations are provided in two formats; DICOM RTSTRUCT contains slice by slice contour points of the external outline of the primary tumour. DICOM SEGMENTATION contains binary masks of the same primary tumour. The nomenclature of the structures are as follows:
- “GTV-1” denotes the index tumour, specifically the Gross Tumour Volume (GTV)
- “vis” denotes manual delineation by radiation oncologists
- “auto” denotes assistance by an autosegmentation tool followed with manual editing by radiation oncologists
- “1”, “2”, …., “5” denotes the individual radiation oncologists working independently of each other
Side note : Radiation oncologists denoted “1” and “3” were trainee radiation oncologists at the time of this experiment. Radiation oncologists “2”, “4” and “5” were extensively experienced at the time of this experiment.
This dataset is intended to be open access to support repeatability and reproducibility of research in the radiomics domain. This dataset has been referenced in Medical Physics Dataset Article addressing FAIR radiomics practices to support transparency, harmonization and collaboration on radiomics (https://doi.org/10.1002/mp.14322).
Other data sets in the Cancer Imaging Archive that were used in the same study published in Nature Communications: HEAD-NECK-RADIOMICS-HN1 , NSCLC-Radiomics-Interobserver1, NSCLC-Radiomics-Genomics, RIDER-LungCT-Seg.
For scientific or other inquiries about this dataset, please contact TCIA's Helpdesk.
Acknowledgements
We would like to acknowledge the individuals and institutions that have provided data for this collection:
- Leonard Wee, MAASTRO (Dept of Radiotherapy), Maastricht University Medical Centre+, Maastricht, Limburg, The Netherlands.
- Dirk de Ruysscher, MAASTRO (Dept of Radiotherapy), Maastricht University Medical Centre+, Maastricht, Limburg, The Netherlands.
- Andre Dekker, MAASTRO (Dept of Radiotherapy), Maastricht University Medical Centre+, Maastricht, Limburg, The Netherlands.
- Hugo Aerts, Computational Imaging and Bioinformatic Laboratory, Dana-Farber Cancer Institute & Harvard Medical School, Boston, Massachusetts, USA.
- Petros Kalendralis, MAASTRO (Dept of Radiotherapy), Maastricht University Medical Centre+, Maastricht, Limburg, The Netherlands.
- Harmonization of the components of this dataset, including into standard DICOM representation, was supported in part by the NCI Imaging Data Commons consortium. NCI Imaging Data Commons consortium is supported by the contract number 19X037Q from Leidos Biomedical Research under Task Order HHSN26100071 from NCI.
Data Access
| Data Type | Download all or Query/Filter | License |
|---|---|---|
| Images and Segmentations (DICOM, 3.2 GB) | (Download requires the NBIA Data Retriever) | |
| Clinical Data (CSV, 2 kB) |
Click the Versions tab for more info about data releases.
Detailed Description
Image Statistics | |
|---|---|
Modalities | CT, RTSTRUCT, SEG |
Number of Participants | 22 |
Number of Studies | 22 |
Number of Series | 64 |
Number of Images | 3886 |
| Images Size (GB) | 3.2 |
Citations & Data Usage Policy
Users must abide by the TCIA Data Usage Policy and Restrictions. Attribution should include references to the following citations:
Data Citation
Wee, L., Aerts, H. J.L., Kalendralis, P., & Dekker, A. (2019). Data from NSCLC-Radiomics-Interobserver1 [Data set]. The Cancer Imaging Archive. https://doi.org/10.7937/tcia.2019.cwvlpd26.
Publication Citation
Aerts HJWL, Velazquez ER, Leijenaar RTH, Parmar C, Grossmann P, Carvalho S, Bussink J, Monshouwer R, Haibe-Kains B, Rietveld D, Hoebers F, Rietbergen MM, Leemans CR, Dekker A, Quackenbush J, Gillies RJ, Lambin P. Decoding Tumour Phenotype by Noninvasive Imaging Using a Quantitative Radiomics Approach, Nature Communications, Volume 5, Article Number 4006, June 03, 2014. DOI: http://doi.org/10.1038/ncomms5006.
Publication Citation
Kalendralis, P., Shi, Z., Traverso, A., Choudhury, A., Sloep, M., Zhovannik, I., Starmans, M.P., Grittner, D., Feltens, P., Monshouwer, R., Klein, S., Fijten, R., Aerts, H., Dekker, A., van Soest, J. and Wee, L. (2020). FAIR‐compliant clinical, radiomics and DICOM metadata of RIDER, Interobserver, Lung1 and Head‐Neck1 TCIA collections. Medical Physics. DOI: https://doi.org/10.1002/mp.14322.
TCIA Citation
Clark K, Vendt B, Smith K, Freymann J, Kirby J, Koppel P, Moore S, Phillips S, Maffitt D, Pringle M, Tarbox L, Prior F. The Cancer Imaging Archive (TCIA): Maintaining and Operating a Public Information Repository, Journal of Digital Imaging, Volume 26, Number 6, December, 2013, pp 1045-1057. DOI: 10.1007/s10278-013-9622-7
Other Publications Using This Data
TCIA maintains a list of publications which leverage our data. If you have a manuscript you'd like to add please contact TCIA's Helpdesk.
Version 3 (Current): Updated 2020/08/31
| Data Type | Download all or Query/Filter |
|---|---|
| Images (DICOM, 3.2 GB) | (Download requires the NBIA Data Retriever) |
| Clinical Data (CSV) |
Resolved the inadvertent mismatch of the labels between the DICOM Segmentations and the RTSTRUCT annotations. Version 2 was replaced.
Version 2: Updated 2019/10/18
| Data Type | Download all or Query/Filter |
|---|---|
| Images (DICOM, 3.2 GB) | (Download requires the NBIA Data Retriever) |
| Clinical Data (CSV) |
Added DICOM Segmentations for the primary tumor only, the ROI (GTV-1) for the RTSTRUCTs and DICOM Segs are the same.
Version 1: Updated 2019/06/02
| Data Type | Download all or Query/Filter |
|---|---|
| Images (DICOM, 2.0 GB) | (Download requires the NBIA Data Retriever) |
| Clinical Data (CSV) |