Summary
This collection contains images from 22 non-small cell lung cancer (NSCLC) patients. For each of these patients with pre-treatment hybrid PET-CT scans, repeated blinded manual delineations by five different radiation oncologist of the 3D volume of the gross tumor volume on CT and clinical outcome data are available. The above was repeated with the same set of five radiation oncologists, using an in-house autosegmentation tool for initial delineation followed by manual adjustment of the gross tumor volume outline. The delineations were cloned to co-registered PET and resampled in the grid spacing of the PET image.
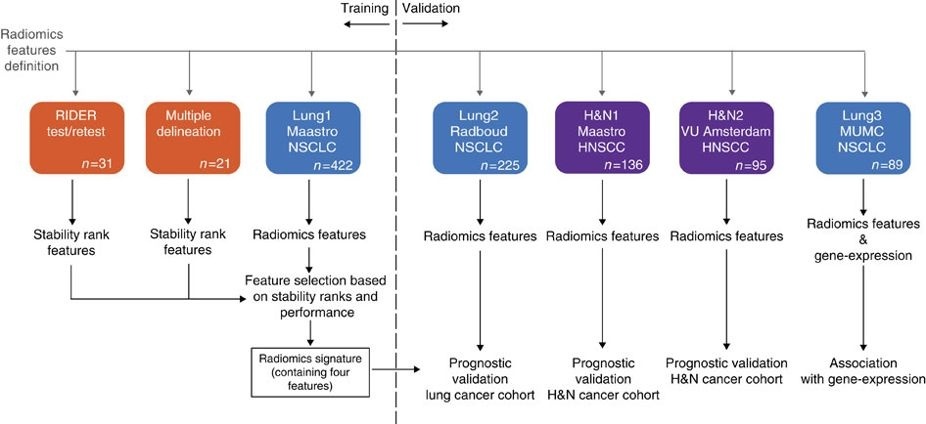
This dataset refers to the Interobserver dataset of the study published in Nature Communications (http://doi.org/10.1038/ncomms5006). In short, the publicates used a radiomics approach to computed tomography data of 1,019 patients with lung or head-and-neck cancer. Radiomics refers to the comprehensive quantification of tumor phenotypes by applying a large number of quantitative image features. In the published analysis, 440 features quantifying tumor image intensity, shape, and texture were extracted. We found that a large number of radiomic features have prognostic power in independent data sets, many of which were not identified as significant before. Radiogenomics analysis revealed that a prognostic radiomic signature, capturing intra-tumor heterogeneity, was associated with underlying gene-expression patterns. These data suggest that radiomics identifies a general prognostic phenotype existing in both lung and head-and-neck cancer. This may have a clinical impact as imaging is routinely used in clinical practice, providing an unprecedented opportunity to improve decision-support in cancer treatment at low cost.
This dataset is intended to be open access to support repeatability and reproducibility of research in the radiomics domain. This dataset will be the subject of an upcoming Nature Data article addressing OPEN, FACT and FAIR radiomics practices to support transparency, harmonization and collaboration on radiomics.
Acknowledgements
We would like to acknowledge the individuals and institutions that have provided data for this collection:
Leonard Wee, Maastro Clinic, Maastricht, Limburg (Netherlands) and Hugo Aerts, Dana-Farber Cancer Institute/Harvard Medical School, Boston, Mass
Data Access
Click the Download button to save a ".tcia" manifest file to your computer, which you must open with the NBIA Data Retriever. Click the Search button to open our Data Portal, where you can browse the data collection and/or download a subset of its contents.
| Data Type | Download all or Query/Filter |
|---|---|
| Images (DICOM, XX.X GB) |
|
| Supplemental Data (clinical image analysis) |
Click the Versions tab for more info about data releases.
Detailed Description
Image Statistics | |
|---|---|
Modalities | PET-CT, SEG, SR, RT |
Number of Patients | 22 |
Number of Studies | 44 |
Number of Series | |
Number of Images | |
| Images Size (GB) |
Add any additional information as needed below. Likely would be something from site.
Citations & Data Usage Policy
This collection is freely available to browse, download, and use for commercial, scientific and educational purposes as outlined in the Creative Commons Attribution 3.0 Unsupported License. See TCIA's Data Usage Policies and Restrictions for additional details. Questions may be directed to help@cancerimagingarchive.net.
Data Citation
Wee L, Aerts H, Kalendralis P, Dekker A. NSCLC-RADIOMICS-INTEROBSERVER1. 2019. DOI: (to be added).
Publication Citation
TCIA Citation
Clark K, Vendt B, Smith K, Freymann J, Kirby J, Koppel P, Moore S, Phillips S, Maffitt D, Pringle M, Tarbox L, Prior F. The Cancer Imaging Archive (TCIA): Maintaining and Operating a Public Information Repository, Journal of Digital Imaging, Volume 26, Number 6, December, 2013, pp 1045-1057. DOI: 10.1007/s10278-013-9622-7
Other Publications Using This Data
TCIA maintains a list of publications which leverage TCIA data. If you have a manuscript you'd like to add please contact the TCIA Helpdesk.